The ACB have also brought in the services of Nirmalan Thanabalasingam as their new physiotherapist
The off-spinner returned for a stint with CSK in 2025
 Indian Astronaut Joins ISS: Shukla's Mission Ushers in New Era for India's Space Program
Indian Astronaut Joins ISS: Shukla's Mission Ushers in New Era for India's Space Program
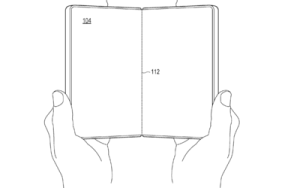 Microsoft Patents Novel Foldable Phone Hinge Design, Eyes Potential Return to Smartphone Market
Microsoft Patents Novel Foldable Phone Hinge Design, Eyes Potential Return to Smartphone Market
 'The Traitors' Star Apoorva Mukhija Accuses Sudhanshu Pandey of Misogyny and Verbal Abuse After On-Screen Drama
'The Traitors' Star Apoorva Mukhija Accuses Sudhanshu Pandey of Misogyny and Verbal Abuse After On-Screen Drama
 Rishabh Pant's Unconventional Batting Redefining Cricket, Says Greg Chappell
Rishabh Pant's Unconventional Batting Redefining Cricket, Says Greg Chappell
 Ashada Gupt Navratri 2025: Unveiling Dates, Sacred Rituals & Hidden Significance
Ashada Gupt Navratri 2025: Unveiling Dates, Sacred Rituals & Hidden Significance
 JPG to PDF: A Graphic Designer's Guide to File Conversion and Quality Preservation
JPG to PDF: A Graphic Designer's Guide to File Conversion and Quality Preservation
 Moto G54 Price Slashed in India: Check Out the New, Lower Cost
Moto G54 Price Slashed in India: Check Out the New, Lower Cost
 England's Audacious Batters Claim They Could Have Chased Down 450 in First Test Win Over India
England's Audacious Batters Claim They Could Have Chased Down 450 in First Test Win Over India
 Van der Dussen to Captain South Africa in T20I Tri-Series Against New Zealand and Zimbabwe
Van der Dussen to Captain South Africa in T20I Tri-Series Against New Zealand and Zimbabwe
 20 Minutes to a Healthier Brain and Heart: Neurologist's Simple Strategies to Combat Cholesterol, Blood Pressure, and Dementia Risk
20 Minutes to a Healthier Brain and Heart: Neurologist's Simple Strategies to Combat Cholesterol, Blood Pressure, and Dementia Risk